An increasing number of people require radiotherapy as an integral part of their cancer treatment.
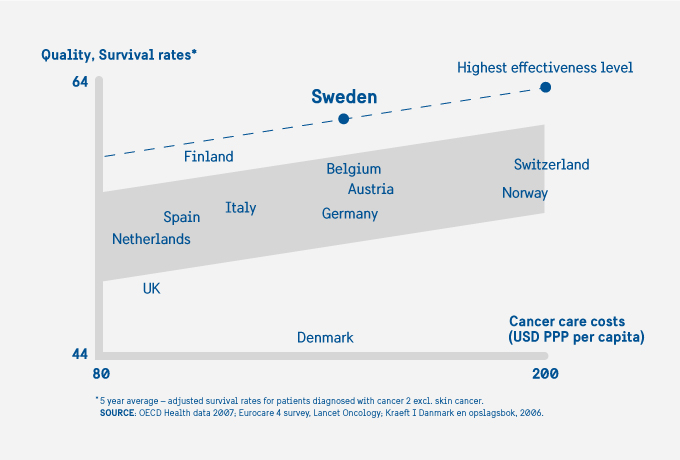
Cancer care in Sweden maintains high standards and stands up well in international comparisons for good outcomes in terms of lower mortality and longer survival. A patient who develops cancer today may live for decades with and after his or her diagnosis. A coherent care process without unnecessary lead times and a good interaction between patient and care provider, based on reliable information and open and trusting communication, is therefore essential to the patient’s well-being and the outcome of care.
Initiatives for a more equitable and accessible high quality cancer care
The health and medical care in Sweden shall be equitable and given on the same terms for all patients, regardless of gender, socioeconomic background or country of birth. Surveys and follow-ups of care outcomes show, however, that there are still unwarranted differences in both health and care between various groups and regions. In response to this challenge, the Swedish Government launched a four-year national program running between 2015 and 2018 designed to make cancer care and treatment more accessible and equitable. A total of SEK 2 billion is being invested in a range of measures with a focus on shortening waiting times and reducing regional differences.
Aim of the Swedish Government’s cancer measures
- Create more equitable cancer care of higher standard
- Improve access by shortening waiting times and reduce regional differences
- Shorten the care process between reasonable suspicion and start of treatment
- Introduce a common national system of standardised care pathways
- Create greater satisfaction among patients through better information and more involvement
These measures are also expected to result in better information, greater involvement and generally more satisfied patients.
Standardised care pathways
The first stage of the four-year program started in 2015 to ensure shorter waiting times in cancer care. The initial aim is to shorten the care process between reasonable suspicion and the start of treatment. To achieve this, the most important measure is the introduction of a national system of standardised care pathways. Besides shorter waiting times, these measures are also expected to result in better information, greater involvement and generally more satisfied patients.
The standardised care pathways describe which examinations and initial treatments are to be carried out for a particular cancer diagnosis, as well which time limits apply – both for the entire process and for the different examination stages. The time limits are to only cover medically justified time and vary between diagnoses and treatments. In 2015, five standardized care pathways were introduced: acute myeloid leukemia, head and neck cancer, esophageal and stomach cancer, prostate cancer, and bladder and urinary tract cancer. In 2016 work will begin to develop standardised care pathways for another 13 types of diagnosis.

The Government has reached an agreement with the Swedish Association of Local Authorities and Regions for the introduction of standardized care pathways and other measures in the four-year investment programme. The National Board of Health and Welfare has been tasked by the Government to monitor and evaluate implementation.
Access to and use of medical technology
Although cancer survival has improved significantly in Sweden, certain types of tumours still have a very poor prognosis . Surgery and radiotherapy are the principal curative methods of treatment. Medicines are used for many cancers, principally as adjunct therapy. Although great progress has been made in the medical treatment of cancer and further improvements will undoubtedly be made, it is preventive efforts that can produce the greatest gains in saving lives and reducing siffering. Early detection is crucial for good treatment opportunities.
In addition to vaccination and screening programmes for common types of cancer, new diagnostic technologies / IT tools will play an increasingly important role in Sweden for accurate detection of cancers such as malignant melanoma and shorter turnaround times for pathologists to determine the correct diagnosis and therapy.
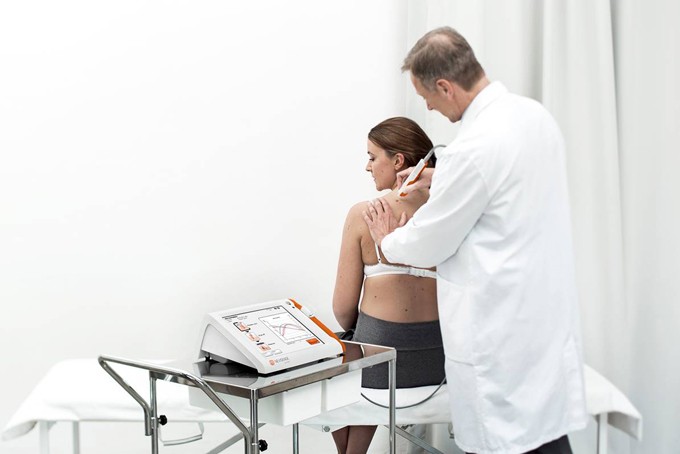
SciBase EIS – Electronic Impedance Spectroscopy – developed at Karolinska Institutet, is a technological breakthrough in non-visual detection of malignant melanoma.
Digital pathology offers unique opportunities for collaborations between hospitals, both regionally and worldwide, as the digital samples can be sent instantly. This is very important in places where, due to a lack of pathologists, the waiting time for the pathology report is often long, with an anxious wait and delayed therapy for the patient being the result.

Digital pathology enables the tissue samples to be analyzed on a computer screen, while digitization of the workflow may result in a better overview of the workload, less administration and shorter turnaround times.
Medicines, medical devices and methods will become an increasingly important element in future cancer care
Brachytherapy solutions are designed for precise, targeted treatment of various cancers such as prostate, breast, skin and surface, rectum and gynecological anatomy. Brachytherapy is suited as a single modality or in combination with other treatments such as external beam radiation therapy.
In Sweden, a rapid development of knowledge in cancer care, medical science and technology is taking place. The increase that has taken place in the survival of cancer patients is linked to improvements in secondary prevention, diagnostics and treatment. As the number of patients with cancer will increase substantially, medicines, medical devices and methods is becoming an increasingly important element in high quality cancer care. All cancer patients, regardless of where they live and who they are, must benefit from evidence-based treatment.
The Swedish Testbed for Innovative Radiotherapy
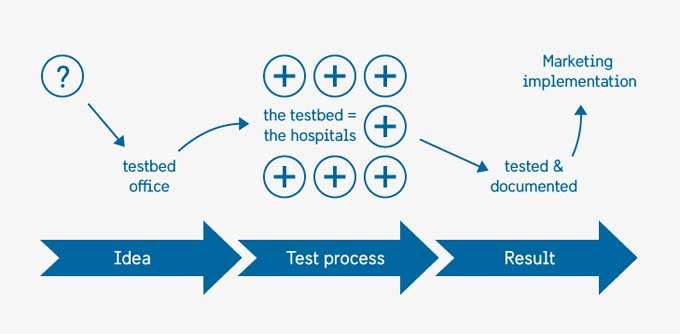
A new initiative for advanced cancer treatment is the highly specialized test bed for innovative radiotherapy and quality assurance. It is also a platform for collaboration between industry and healthcare providers. The testbed is used for developing products, services and organisational solutions that are needed in the healthcare sector. Companies test their innovations in relevant environments and can shorten their development process while care providers have the possibility to influence and quicker implement innovations. Academia benefits from access to the most modern technology for conducting multicenter studies in clinical settings.
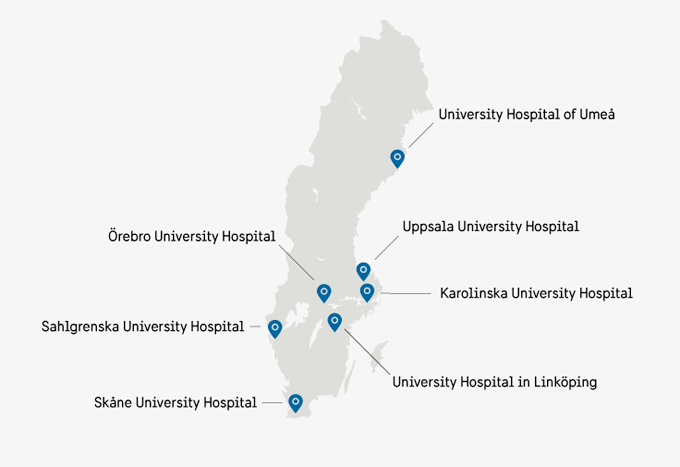
The collaboration platform is a joint investment by healthcare and industry. Partners in the consortium are all seven Swedish university hospitals, six specialist medical technology companies and the six regional cancer centres. The investment is being co-financed by Vinnova, the Swedish innovation Agency and is coordinated by the Innovation Centre at Karolinska University Hospital.
Current ongoing projects within the testbed are MRI-based radiotherapy, quality assurance for MRI-based radiotherapy, adaptive radiotherapy, patient positioning and immobilization and the development of a national database solution for comparable radiotherapy data.
This highly specialized national test environment is not only a win/win situation for the medical technology industry and healthcare providers. It also adds value for the cancer patients through the possibility to access and to contribute to research and development of evidence-based, effective and safe cancer treatment.
Photo credit: Elekta, SciBase, Sectra, Alexander Ruas/Capio